
All journeys in implant dentistry start with aquiring “primary” stability, which is nothing but static friction between the newly placed implant and the bone. But nothing good comes without pain and no friction without bone compression..!
Starting from this basic fact, in this article we disect our common perceptions about primary stability, bone resorption, aggressive and passive implants and we finally end up introducing a new term, busting one of Nikos’ oldest slides and opening up much more for discussion..!
… meet Friction Stability..!
Achieving the essential “primary stability” has been the starting point in every implant journey, yet it has been something notoriously difficult to define. Some clinicians have been looking for a certain value of insertion torque, researchers often defined it as a threshold of micromobility, while some few have actually questioned whether primary stability is truly essential for osseointgration (1). In clinical settings however, most of us will aim for an implant that is “clinically stable”, meaning it does not present with any visible movement and cannot be spinned in the osteotomy with finger torque.
This initial stability is purely due to static friction, the mechanical interlocking of the implant with the surrounding bone. The higher the friction, the higher the insertion torque and the stability we “feel” in clinical terms. “Friction Stability” might have actually been a much better term to use for what we observe after insertion. And if you remember from your high school physics, the friction between two surfaces is proportional to the force pressing them together. In our case this is the force applied by the implant to the bone, or the bone compression.
Common sense Lesson Nr. 1: there is no such thing as a “passive” dental implant, which also can give primary stability. If your implant is clinically stable after placement, then it does compress the bone somewhere.
– Now where exactly, would be worth some further discussion.
How do implants achieve enough friction to give clinical stability? No rocket science here again, mainstream implants use the two oldest tricks in the book (of mechanics): threads and “wedging”. Threads are typically wider than the osteotomy so that they “bite” in the walls of the osteotomy and stabilize the screw. Wedging means having an implant that is slightly wider than the last drill consequently pressing as a “wedge” the bone walls and increasing in this way the friction. In the past some true wedge-form implants were marketed, such as the Endopore which had no threads and were literally hammered in the osteotomy. They achieved primary stability and osseointegrated, though I am not sure of the success rates. The idea (or the company) eventually died out.
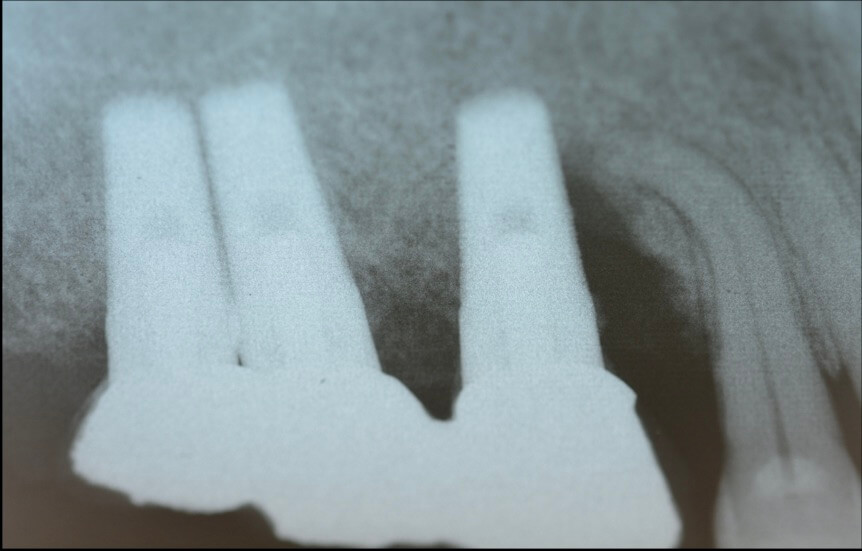
Bone Compression… friend or foe?
So is compression bad for the bone? Not necessarily!
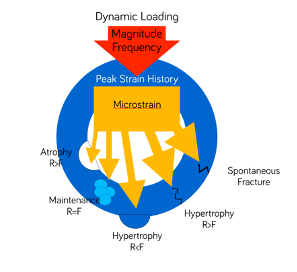
Nobody likes pressure, but mechanosensitive tissue like bone has actually evolved with primary objective to respond to pressure. The classic work of Frost (2) on the Wolf’s law has shown that forces of certain intensity will actually have an anabolic effect on the bone, while it is only when forces exceed a certain threshold that they become detrimental. (Does this sound a bit like the common “clinical wisdom” that some insertion torque is good, but too much might be more trouble than benefit?)
Although Wolf was able to calculate this threshold of strain for long bones in humans, we really know very little of how this fundamental principle would apply in the micro-structural environment of the complex alveolar ridge.
The actual bone response of different segments of the alveolar ridge to different levels of forces applied by an implant remains unknown. One can only assume that force beyond the elastic modulus of the bone will cause plastic deformation, commonly known as fracture and possibly necrosis, if the bone segment is squeezed beyond its repairability or deprived of vascularization
…but then why this wide-spread perception that compression kills bone?
What we know about the bone response to compression comes from some classic histology studies from the Gothenburg team of Lindhe, Lang, Berglundh and Abrahamsson (3). The team placed implants in dogs and conducted histological measurements at certain time intervals thereafter, looking closely at the histological progress of osseointegration.
The “big deal” however was on the implant they used: It looked like Straumann, but it was not.

Not only it had very protruding threads, but there was an additional “pitch” at the top of each thread which would have probably made the traditional Straumann users of that time scream in disgust! So this implant never made it to humans, but remained in the notoriously small market of Beagle-dog implants. Nevertheless, this design was genius for the purpose of the experiment, as it created areas of compression (at the pitch of each thread) and areas of no contact, or empty chambers in the surface between two threads.
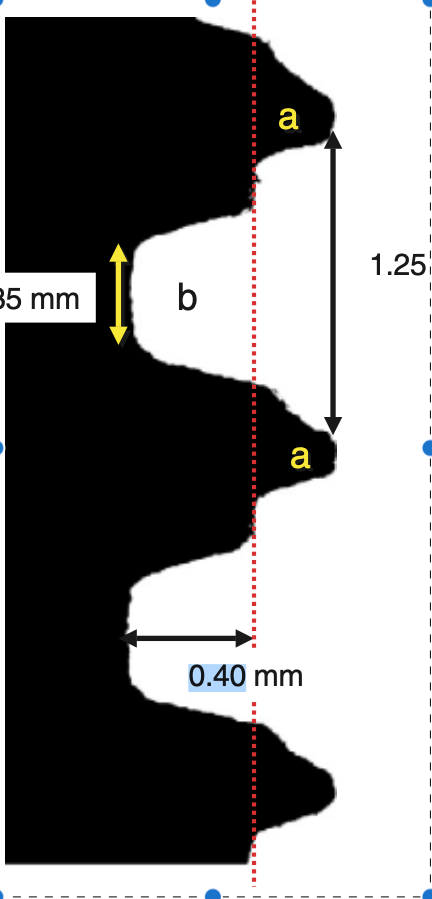
Right after implant placement, friction stability was achieved thanks to the pitches of the threads biting tight into the bone walls, while the non-contact areas filled with blood clot. The big news came however on the 4th day, when an interesting pattern of bone resorption occurred at the coronal side of the pitch! Why resorption? Obviously the compression of the bone there had killed a zone of bone about 50 μm, which was now being cleared away.
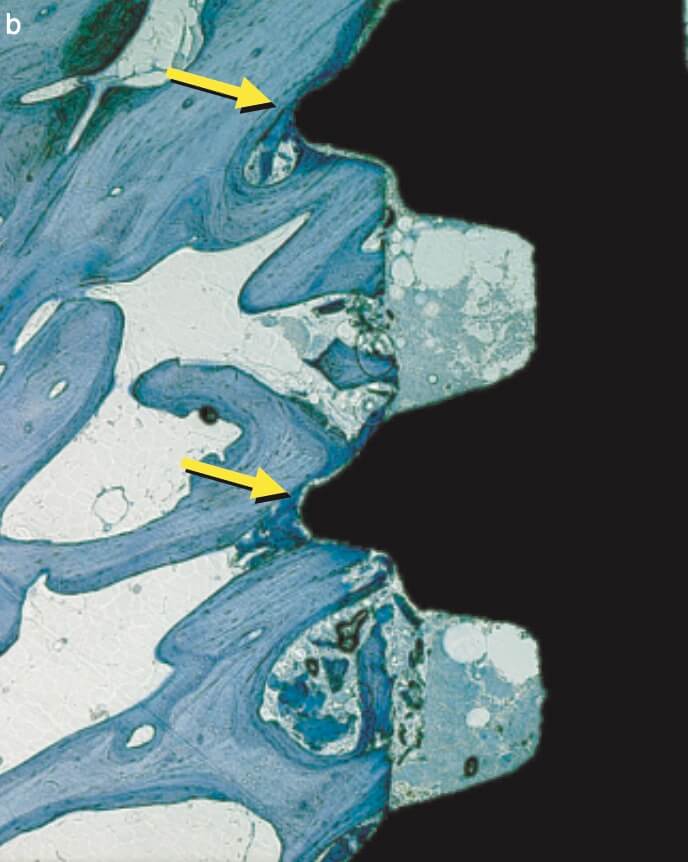
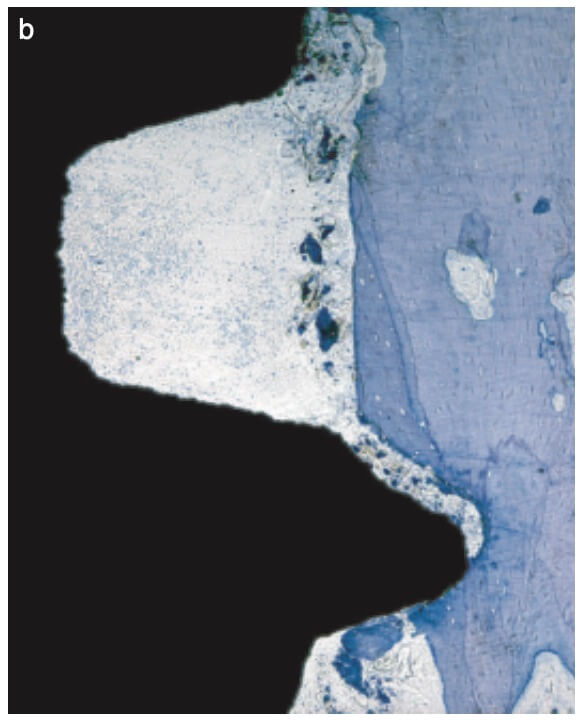

And why the top of the pitch and not the bottom? Well, this is actually screw mechanics. Regardless if you work with implants or with IKEA furniture, screws only apply force through the side of the thread which faces their side of insertion. A screw thread has actually only one working side. We learned this well when we were slicing implant-abutment junctions, only to see how little surface of the actual screw is engaging with the implant and always only the coronal part. (We learned a lot by looking at screws that time, but that would be probably a topic for another post). Common engineering never ceises to amaze!
Back to our histology study, the observation of the 4th day was actually an “aha” moment for all. It showed how compression kills bone and initiates resorption early after placement. This observation seen however in isolation made many clinicians to blame extensive threads as “bone killers”, starting the perception of implants with shallow or few threads as being “passive”. (It was also probably the reason why this experimental implant remained forever a dog-implant).
– But, like always, the complete truth is more complex..!
… meanwhile, in the empty chambers…
To get the whole picture, we need to look closer at the other side of the coin and see what happens in the no-contact areas. There, at 1 week one could see another exciting development: contact osteogenesis! New mineralized bone was created on the surface of the implant in the empty chamber, where previously only blood clot existed. This was something hardly seen on the smooth surface implants, but was apparent in the SLA implants introducing another factor in the equation, the implant surface.
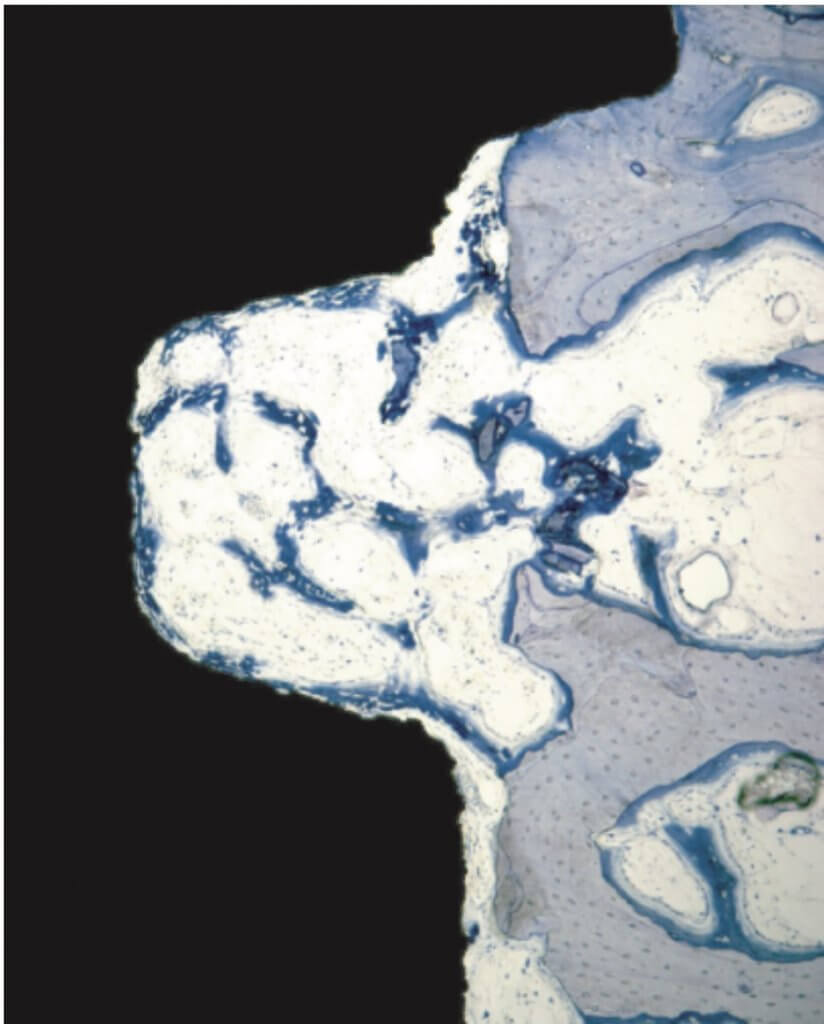
The collective message of these amazing experiments was the while bone is being cleared at the pitch of the thread, new bone is getting created on the implant surface at the no-contact areas! Surface is a significant factor here, with contact osteogenesis being well established at 2 weeks in humans with SLAactive implants, as documented by another great study by Lang et al (4). Lang as well, noted absence of new bone at the pitch of the thread after 1 week, but significant new bone in non-contact areas, where the “jumping” distance between the implant and the bone was short. Again, this message seen somewhat superficially, lead many to believe that the lower and fewer the implant threads, the better the implant would be, reinforcing the anecdotal concept of “aggressive” and “passive” implants. But as you can’t judge a book by its cover, you shouldn’t also judge an implant by its threads! The missing part here was that an implant with few and shallow threads will be unlikely to offer adequate primary stability!
Lesson Nr. 2: you can’t judge a book by its cover and you shouldn’t also judge an implant just by its threads!
At this point, some might question how do we get primary stability with tissue level implants then, having so few and mild threads? Again, the two oldest tricks in the book: if you don’t make use of the threads you will have to use the wedging, or most likely a combination of both. Indeed, the tissue level implants are 0,6 mm wider than the last drill, which could create a significant bone compression effect, not just gathered at the threads, but most likely spread throughout the whole body of the implant or the most narrow part of the osteotomy. So yes, what many perceived as a definition of a passive implant, might not be that passive after all! Maybe borrowed from psychology “passive aggressive” would be a better term..!
In the case of the Tissue Level implant, many of my friends have argued that bone compression is reduced significantly thanks to “tapping” before implant placement, something that I have however to disagree.
Tapping will reduce insertion torque of the implant, but not the bone compression. Tapping is cutting the shape of the implant in the bone, using a “thread cutter” which has exactly the same dimensions as the implant. This way it clears out the 0.6 mm jumping distance compressing the bone, parts of which will get a plastic deformation as a result. So when the implant comes, the bone is already pushed out of the way with the segments under plastic deformation offering no longer friction, while those under elastic deformation will contribute to stability pushing back on the implant surface. The same will happen if you reverse the implant a few threads and then torque again, as many clinicians will do when the insertion torque gets too high. Taping or tricks such as reversing will reduce insertion torque, but the overall bone compression will be the same.
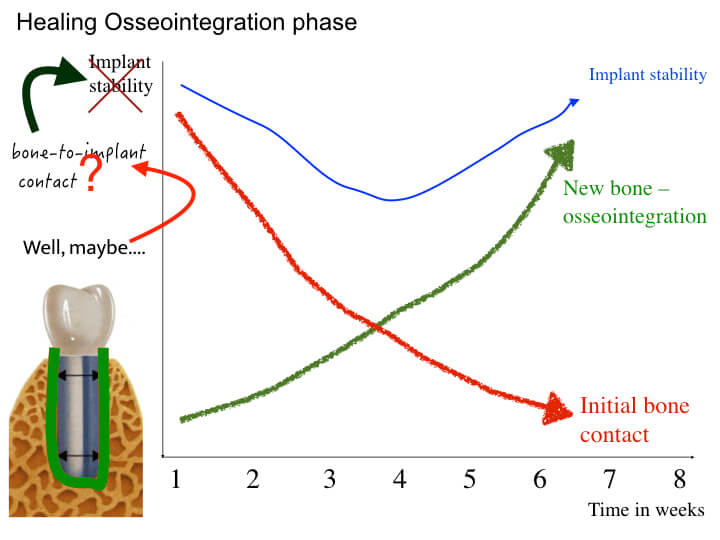
I think it’s time to review some of our oldest and possibly strongest perceptions! Do you remember the classic graph of how gradually primary stability turns into secondary, leaving a sensitive window of stability dip in-between? A classic, isn’t it?
It was for me too. It actually is one of my slides in the “Introduction to Osseoinregration” I have been giving since I started teaching Implant Dentistry. Until I suddenly realized that this graph was a pure and shameless speculation, based on assumptions deriving from interpretations of bone-to-implant contact in histological studies. How this really translates to implant stability and how this famous “dip” occurs -or doesn’t – is something that has actually never been shown in clinical studies (actually, not even animal studies). Things might just be more complicated… Well, with apologies to all my past students, to the left you can see the currently updated version of the slide, as it would be shown in the seminars from now on..!
So, to make a long story short…
…back to square one, what happens between friction stability and osteointegration and what is the role of the implant design and surface in this process might be something far from clear. The interplay between osteotomy protocol and drilling sequence, the implant design with areas of compression and areas of no-contact, as well as the implant surface might be a key to unlock new individualized implant placement and timing/loading protocols. But this post is getting long already and this is only the start of the discussion..! How do all these translate to some implant design and clinical implications? I guess myself and Francisco have some work in progress there, so let us all come back to discuss this next time 🙂
Learn the latest news first, subscribe to my newsletter!
References
- Szmucler-Moncler et al 1998. Timing of loading and effect of micromotion on bone-dental interface: a review of the experimental literature. J Biomed Mater Res 1998.
- Frost HM. A 2003 update of bone physiology and Wolff’s Law
for clinicians. Angle Orthod 2004;74:3–15. - Berglundh, Abrahamsson, Lang, Lindhe.
De novo alveolar bone formation adjacent to endosseous implants. A
model study in the dog. Clinical Oral Implants Research 2003; 14: 251-262 - Lang NP, Salvi GE, Huynh-Ba G, Ivanovski S, Donos N, Bosshardt DD.
Early osseointegration to hydrophilic and hydrophobic implant surfaces in humans. Clinical Oral Implants Research 2011; 22 (4): 349-356.
22 thoughts on “From Friction Stability to Osseointegration and all things in-between..!”
Great content! Super high-quality! Keep it up! 🙂
Very thought provoking read! Many thanks!
Much interesting and really informative .expecting more…many thanks for sharing
Really? The unified theory of osseointegration? You Nick.. are the Einstein of Implantology… (With due apologies to the late Alfred)
haha, it was a (not so successful) joke! I hope Albert will forgive me 🙂
Much informative and systematic than best of scientific implants articles. It may open doors of osteogenesis and or osteogenic remodeling which may have paradigm change in periodontal regeneration.
Thanks for sharing
Thank you for your kind feedback and insight Dr. Deepak!
Very smart argumentation on scientific literature. I love your way to share perio/implant knowledge beyond the dogma!
evet çok yararlı olduğunu düşünüyorum
Since 1985 plateau and locking taper implant had friction stability without screwing the implant in the bone.
this is the unique implant that have haversian and functional perimplant bone.
only in this type of bone we can apply frost and Wolff law.
so we have in this design real clinical short implant and in others only market short implant.
its good to explain this because the mayority of dentist dont know the real bone perimplant physiology
thanks
Dear Andrea, Many thanks for your feedback and insight!
Have you ever thought about including a little bit more than just your articles? I mean, what you say is fundamental and everything. Nevertheless imagine if you added some great graphics or video clips to give your posts more, “pop”! Your content is excellent but with pics and videos, this blog could definitely be one of the very best in its field. Awesome blog!
I am really grateful to the owner of this site who has shared this
wonderful article at at this place.
Hi! I just wanted to ask if you ever have any trouble with hackers?
My last blog (wordpress) was hacked and I ended up losing a few months
of hard work due to no backup. Do you have any solutions to prevent hackers?
Long time supporter, and thought I’d drop a comment.
Your organization’s wordpress site is very sleek – hope you don’t mind me
asking what theme you’re using? (and don’t mind if I steal it?
:P)
I just launched my small businesses site –also built in wordpress like
yours– but the theme slows (!) the site down quite a bit.
In case you have a minute, you can find it by searching for “royal cbd” on Google (would appreciate any feedback) – it’s still in the works.
Keep up the good work– and hope you all take care of yourself
during the coronavirus scare!
~Justin
Hello Justin and thank you for your kind comments and feedback! The theme is Academia, with some minor customizations, as I am still learning how to use wordpress. It is a very powerful tool, but being an amateur there is a lot to learn and little time. My blog in the past was run on Moodle, which is also a great tool, but as Php was advancing I was left behind and could no longer find time to catch up… WordPress seems easier to maintain. I will check your site at first opportunity! best regards, N
great article , got answers to some eluding questions
great article , got answers to some eluding questions
Wow! Very informative. I wonder if you only write about implants and dental topics or you have your dental clinic?
Thank you for your kind comment! yes, of course, I am first and foremost a dentist as well, currently practising in Hong Kong.
Wow! Very informative. I wonder if you only write about implants and dental topics or you have your dental clinic?
Thank you for your kind comment! yes, of course, I am first and foremost a dentist as well, currently practising in Hong Kong.