Tissue level versus Bone level implant is a discussion that goes back to the very DNA of implant dentistry. Although both designs have peacefully (more or less) co-existed for decades, from time to time the discussion reshuffles every time some new data is published, or a new product hits the market!
Today we will scrutinise the evidence and history trying to respond to a simple emerging question: “Does the use of tissue level implants reduces the risk for peri-implantitis?”
by Nikos Mattheos, DDS, MASc, PhD
In the beginning, there was one implant…
… this implant was Brånemark and it was a “Bone Level”, in the sense that is it was meant to be placed in the bone in its entire length. Looking at the first Brånemark implant serries today, one might be misled by its cylindrical top, the “platform”, which clearly sticks out from the screw threaded part of the implant. The intention however was always to place the implant at the level of the bone, if not actually somewhat submerged under the bone. There was a simple reason for this in Brånemark’s original paradigm: the implant was always meant to be healing submerged, hidden under a thick layer of soft tissues. This is understandable, as during the pioneering years of Implant Dentistry, Brånemark was aiming to isolate the implant as much as possible from the oral environment and secure immaculately aseptic conditions for the surgery.
If you ever come across his first textbook, take the time to read the chapters for patient selection, preparation and surgical set up. It is astonishing to see the pedantic level of detail for asepsis in his surgical set-up protocols, including my all-time favourite, the dressing up of the surgeons in astronaut suits, equipped with a closed breathing circuit, so that not even the air they breathe will come out to contaminate the surgical field!
Ironically, today, many decades later, implant surgeons started to dress up again like in the Brånemark times, but this time for a whole different reason…


The first implant was a… bone level!
The Brånemark implant with flat to flat, external hex connection.
No, it’s not Covid, it’s one of the pioneers from Brånemark’s team preparing for surgery !
…then another implant came..!
…and this one looked very different. It was the implant designed by Andre Schroeder in Switzerland and came out a few years after Brånemark’s. What was very different in this implant was the “toulip” shaped collar on top of the threaded part, which however was designed to “stick out” of the bone and provide the space for the soft tissue attachment.
No, I don’t think it was peri-implantitis that Schroeder had in mind when he designed his “tissue level” implant, his motivation was actually very practical. Unlike Brånemark, Schroeder was proposing transmucosal healing, that means allowing the implant to heal through the soft tissues, while being from the beginning visible in the mouth and exposed to the oral bacteria. Thus, the transmucosal “collar” would help in the stabilisation and adaptation of the soft tissues around the implant. Today, transmucosal healing is a standard procedure, but back then it was a very bold move, which actually took many years to gain acceptance from the wider Brånemark users. Actually, thinking of the imprecision of implant prosthetic components
and junctions in the early period of of Brånemark, transmucosal healing would have only been possible with a one-piece implant configuration.From that time, two different paradigms in Implant Dentistry emerged, both with loyal fans and devoted supporters, as well as fierce critics. Brånemark’s paradigm morphed into Nobel Biopharma and later Nobel Biocare portfolios, while Schroeder’s gave birth to the ITI and Straumann implants. The question of Bone Level or Tissue Level back then was mainly responded depending on which of the two camps one subscribed to and for decades there was little overlap between the two paradigms.
In the late 90’s during my training in Sweden, I had the rare luck to witness some discussions between ITI and Brånemark users, which today would have sounded more like exchange of arguments between football fans! But so it was back then, excitement (and emotions) were running high. It took Straumann a couple of decades to acknowledge that Bone Level implants can have advantages, while it took Nobel Biocare even longer to allow the conversion of their implants into Tissue Level with a “one time” abutment, effectively acknowledging that tissue level can also have benefits.

The implant by Schroeder, known as ITI, Bonefit or later Straumann. The endosseous part of the implant was supplemented with a “Toulip” shaped collar on top for the soft tissue attachment, while it included an internal connection.
and what about now?
Today, we live an era of convergence, where successful practices and designs are quickly exchanged throughout the spectrum of implant dentistry and implant design portfolios start at times to look dangerously similar across the board. It’s been a long time since I last heard people fighting about implant designs, which is certainly a good thing and it also means that we can now look into the facts with a clearer mind.
Although today the question of “Tissue or Bone level” would have a much different purpose than back then, it remains a valid question which tends to surface again every time a study brings up some new data. So allow me to tackle this old question in the light of the latest data and paradigms.
There are many valid reasons for someone today to choose one over the other, and I will not attempt any comprehensive comparison, as this would take too much writing (and honestly, I hardly qualify to cover all aspects). Nevertheless, I believe the design of the Implant Supracrestal Complex is a critical issue in modern implant dentistry. These few critical millimetres of the reconstruction, from the bone margin to the top of the soft tissue will determine to a large extent the longevity of the prosthesis in healthy tissues. Therefore, the choice of tissue or bone level is important and should be directed by the individual conditions and the design we aim for. You can read more in some of my older posts.
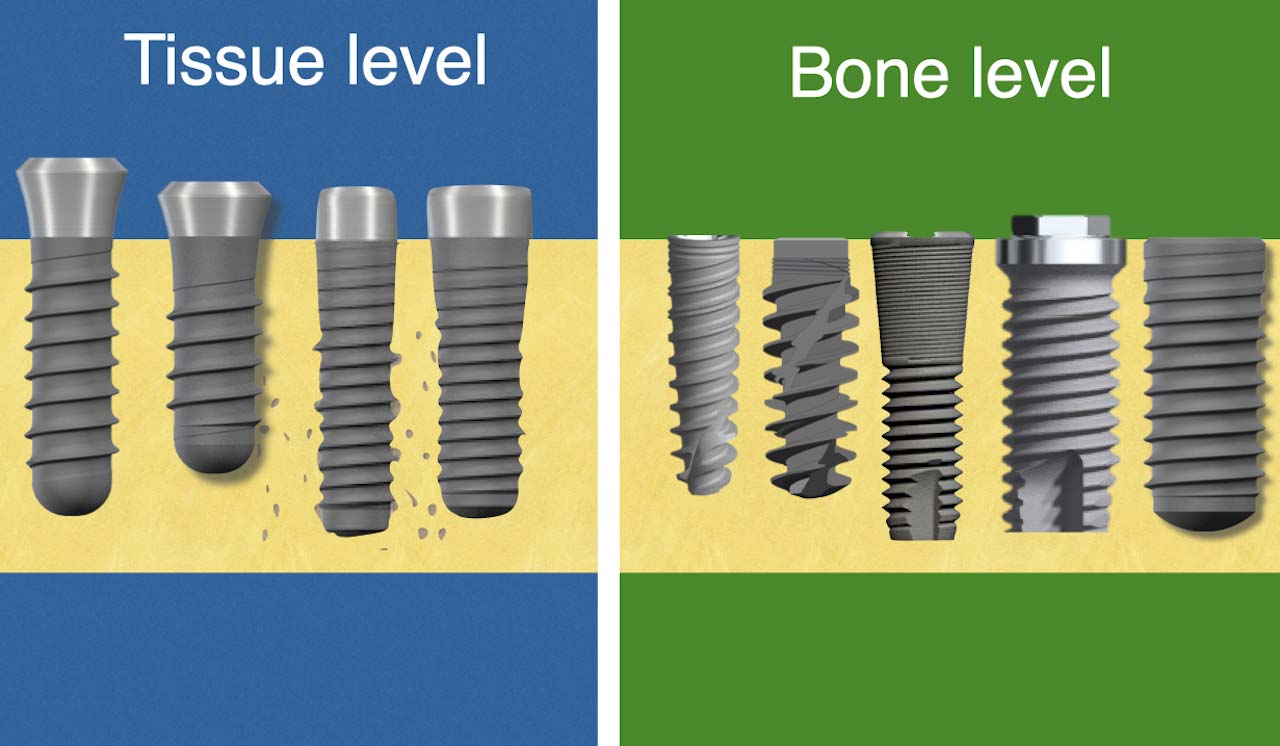
Tissue Level vs Bone Level implant:
A (soft-) Tissue Level or often called "one piece" implant has a polished "collar" on top of the rough surface, which sticks out of the bone and facilitates the attachment of the peri-implant soft tissue. A bone level implant on the other hand is meant to be fully submerged under the bone and soft tissue attachment is facilitated by an extra piece, typically a screw retained abutment.
Today I would like to discuss something more specific and that is would the choice of Tissue or Bone Level implant affect the risk for peri-implantitis?
Tissue Level implants have been since long the “darlings” of old school periodontists, like myself. I have also seen official recommendations in the past suggesting the use of Tissue Level implants in patients with history of periodontitis. Placing this question on a social media post could trigger a vivid discussion and exchange which lasted for a week, almost like the good old times..!
The split of opinions in this question is intriguing and an indication that the old paradigms and the DNA do not simply vanish, but morph into deeper layers in all of us. There seems to be a true belief that tissue level implants reduce the risk of peri-implantitis among many colleagues, as much as this is questioned by others and all suggesting evidence for this in studies.
- But in an era when you have to constantly prove that you are not an elephant, would this belief stand the test of evidence? What is the evidence we have to support this? In short, which study showed that tissue level implants do reduce the risk of peri-implantitis?
the short story is…
…None. Or better, none I could find evidence for. And that was the short story, you can stop reading now if you are bored. Or you can continue for the long story as well.
the long story
I have been trying to scrutinise the literature looking for studies out there which were designed to answer this simple question: “Do tissue level implants reduce the risk of mucositis and peri-implantitis when compared to bone level implants?” And – surprise-surprise- I could find one!
It was a split mouth, randomized, prospective study, conducted by Vianna, et al. (2018). They took 20 patients with history of Periodontitis and restored each of them with one Tissue Level and one Bone Level Straumann SLA implant. Both implants were placed at posterior sites in the same surgery and the final decision which would be TL and which BL was made through randomisation with sealed envelopes. Both implants healed transmucosally and were restored with Metal-Ceramic, screw-retained single crowns. Patients were followed up for 2 years and what was the difference? No difference. Not only there was no difference in diagnosis of Mucositis or peri-implantitis, but there was no difference in any clinical parametres: Plaque scores, Bleeding on Probing or Probing depths. And this is where the story ends, leaving still much to anticipate, but this is as much solid evidence as we have, because this is the only study I could find, which was actually designed for – and thus able to respond to – the question we posed at start. If you find anything better, please, surprise me!
A small disclaimer here: we were looking for impact on mucositis and peri-implantitis, that means plaque induced inflammation of the peri-implant tissues. To show this you need clinical examination, bleeding on probing, probing depth, redness, swelling, suppuration. Any study without such data simply would not do. Marginal bone loss at 6 or 12 months or more, especially when assessed by unstandardised Periapical radiographs is necessarily not peri-implantitis. Actually, I don’t know really what it is and if we should worry about it. It depends. But for now, back to peri-implantitis.
the even longer story
Could it be that this is all? There must be some other studies that show tissue level to be superior in terms of peri-implantitis risks, I’m sure you heard of them, haven’t you? Well, that’s all when it comes to studies which are designed to address this question. (Again, we are talking about peri-implant tissue inflammation, not simply some sort of “bone remodelling” especially when all we have to measure it is unstandardised PA radiographs).
But there are some retrospective and cross sectional studies, which have touched upon the tissue level – bone level theme and have prompted some conclusions. What is however the main problem of such retrospective and cross sectional studies? They are not designed to seek differences between tissue and bone level implants, they merely report numbers they came across while looking for other things. So with regards to TL/BL comparisons, the first direct compromise is a rather huge selection bias. This means that someone at some point made the choice of TL or BL based on his or her own perceptions and philosophy, his own bias. As different operators will use different systems, others will actively select bone level or tissue level implants for different indications, any opportunistic cross sectional or retrospective comparison is deeply problematic. But even taking aside the bias, there is not much to see in terms of difference, if we look closer at these studies.
For example, many colleagues have used the recent study by Katafuchi, et al. (2018) as the evidence that Tissue level implants are less prone to peri-implantitis. Katafuchi et al published a very interesting study in many ways and it is true that they reported different numbers in peri-implantitis prevalence in TL and BL. They found peri-implantitis at 22.8% implant level / 28.9% patients with bone level implants versus 7.5% implant level / 14.8% patients with tissue level implants. Big difference, don’t you say?
But no, not really… the authors report different numbers, but they reported no difference. How can this be possible? Well, it’s as we said the limitation of the sample. The study was obviously not designed to respond to this question, the authors were looking for different primary outcomes. The sample composition is not described in detail, but based on a reference provided to a previous study (Daubert, et al., 2015) it can be concluded that it includes implants of 8 different types/brands, with and without platform switching, with screw-retained and cemented restorations, immediately loaded and grafted sites. What is then defined as “bone level” includes a big diversity of configurations, which makes it very difficult to identify which element of the design is the critical parameter related to any observation. There are major differences among tissue level implants as well, in terms of collar height and angle, even within the same brand. With the study being retrospective and so many confounding factors, the authors did not attempt any statistical comparison between the peri-implantitis prevalence in Bone and Tissue level implants and in my opinion, they did so very wisely. So, no, this study does not show a difference in peri-implantitis risks for TL and BL.
Likewise, Yi, et al. (2020) in another retrospective study of similar design, reported different prevalence in peri-implantitis with Bone Level external, Bone Level internal and Tissue level showing 29.8%, 17.5% and 13.6% respectively. Again, the sample included 8 different types/brands of implants, screw-retained and cemented restorations, single and splinted prostheses, immediate and staged placement, 1 and 2-staged protocol and augmented sites. On top of the previously discussed confounding factors, this study also had a relatively small number of tissue level implants. Nevertheless, statistics were attempted and there was no significant difference found. So, again, Tissue level or bone level, no difference.
After that, it gets even more straightforward. Monje, et al. (2018) in another retrospective matched case-control study found no difference in Bleeding on probing, Mucosal Redness and Probing depth between Tissuel Level and Bone level implants, while Rokn, et al. (2017), in a cross-sectional study found again no significant difference in the prevalence of Mucositis and Peri-implantitis between Bone and Tissue Level implants. And the long-term cherry for the pie comes from Chappuis, et al. (2016) who in a retrospective study of 5-9 years found no difference between TL and BL Straumann implants with regards to Suppuration, Modified Plaque Index, Modified Sulcus Bleeding index and Probing Depth. Are we missing something?
Well, an honorable mention to a great study by Jan Derks et al. In a ground-breaking study some years ago, Jan Derks concluded among other things that there was a significant difference in the late implant loss between implants of different systems, with Straumann showing the lowest. The fact that these implants were mainly placed in the 90s up to 2004 and before Straumann came with a Bone Level implant, prompted many to attribute this difference to the superiority of the Tissue Level as opposed to the Bone Level. To do this however, would take the speculation to whole new level. First of all, peri-implantitis and late implant loss are two very different things, with only connection that the first can at times lead to the latter. Then there is an endless list of parametres here, making this a playground for any hypothesis possible. For example, knowing how the implant practice was in Sweden in the 90s, almost certainly a large portion of the Nobel Biocare implants (if not the great majority) in this sample must have been of the Brånemark series. This could mean internal connection vs external. It could also mean full arch vs smaller bridgework and crowns. My own personal favourite hypothesis is actually behavioural: at that time the ITI philosophy and thus the main training of the Straumann users was very different to the mainstream views within Nobel Biocare users. It was a time when many key opinion leaders of N/B questioned the very existence of peri-implantitis, with a well spread attitude against probing around implants, which was deemed unnecessary if not damaging…
Again, it’s my speculations against these of anybody else, so feel free to pick what you like best!
Conclusively, there is today valid evidence at least with regards to short-medium term, which shows that there is no difference in terms of the risk of Mucositis and Peri-implantitis between TL and BL. You might say two years is too short for peri-implantitis, but certainly not for Mucositis. Now with regards to the long term, the retrospective studies available do not show any difference, but cannot exclude the possibility either. You can still believe that tissue level implants will reduce the risk for peri-implantitis if you chose to, but you will certainly have a hard time to find convincing evidence for this.
Should there be a difference?
This is now the difficult part. Why difficult? Because here we cross the borders of evidence (even weak) and we enter the domain of pure speculation. There is nothing wrong with speculation, actually I do it all the time. It is only through hypothesising that we move forward, as long as we can clearly distinguish when we talk about evidence and when we speculate. And we all speculate based on our paradigms, which means that our hypotheses are typically inclined towards proving what we believe and sometimes disproving the beliefs of others. It’s cognitive dissonance and it’s usually fine, as long as you admit it. So I will continue these lines with a very clear understanding that my speculations are no better – or worse – than anybody else’s. Why should there be a difference between Tissue and Bone level with regards to risk for peri-implantitis? The difference between the two is that in the Tissue Level, the implant-abutment junction is moved 2-3 mm coronally, from near the bone margin to somewhere in the peri-implant sulcus. To some this was enough to justify a protective function of TL: it takes the junction away from the bone. To me, this would be exactly the opposite: it takes the junction in the sulcus! Peri-implant tissue inflammation does not start in the bone, it starts in the sulcus and actually the top of it.
There are three ways I can see a potential impact of the implant-abutment junction towards peri-implant tissue inflammation: Abutment mobility, Microleakage and Plaque retention. You can get a deeper insight on how implant-abutment junction can predispose to complications, in our Masterclass on the management of Implant Complications.
Abutment mobility and microleakage are kind of interrelated, as they both depend on the tightness and the fit of the connection. Although in the early days,
connections in general suffered from significant imprecision, the implants we use today have to a great extent resolved this issue. The fit of the modern internal connections, especially in morse taper parts is impressive and goes down to microgaps that are less than 1-2μm. Unless screw loosening occurs, I see little chance for mobility or micromovement having an impact in both types.
Plaque retention is possibly the most interesting, as to my understanding a junction in the sulcus (as in the TL) is much more likely to become plaque retentive, than a junction under the Junctional Epithelium, as in the Bone Level. Plaque accumulates in the margins of the prosthesis which is exposed in the mouth. If not removed, inflammation starts from the top of the sulcus first, then expands. A stable implant-abutment junction which is 2 mm apical of the Junctional Epithelium cannot have an role in Mucositis which starts at the top of the sulcus. On the contrary, a junction like the one at Tissue Level implants which is in the sulcus, would be much more prone to plaque retention, if biofilm reaches the significant undercut between the abutment and implant collar, which in Tissue Level implants is about 50μm. So if you ask me, a Tissue Level implant placed in a site with thick soft tissues (deep sulcus) would be much more risky for peri-implantitis than a bone level one. Add to this a potentially low collar height, such as in TL implants with 1.8 mm collar and I see a serious contraindication for the aesthetic zone for example. This is one of my observations in the study by Chen et al, where crowns on deeply placed TL implants had to be removed and cleaned outside the mouth in order to get rid of established Mucositis, as the biofilm had reached the implant abutment junction. Disinfecting the implant from biofilm beyond the implant abutment junction, is a whole chapter in itself and you can read more about it in our “tips & tricks” section.
But then again, if the prosthesis is cleanable and the patient can clean it, does it really matter where a stable junction is located? I think not.


The implant-abutment junction area (green circle) on a tissue level implant (left) and a bone level implant (right) under the SEM. The dotted line represents the gingival margin, if it is assumed to be 3mm coronal of the bone. The implant-abutment junction is coronal of the junctional epithelium but in the sulcus in the TL, but apical of the Junctional Epithelium in the BL.
The same area in larger magnification (65x).
The junction is very tight for both implants (<2μm), but observe the undercut area of > 40μm in the tissue level implant, which is about 1mm below the assumed soft tissue margin in this case. Which of the two is more likely to become a plaque retention point?
(Images from Mattheos et al 2016, Fokas et al 2019)
So where are we now?
We are in the great position which I think both Brånemark and Schroeder would have loved: we are able to say that Bone as well as Tissue Level designs are both very successful configurations that can bring predictable and sustainable treatment outcomes. They both have indications and they both can be part of our treatment array. The fact that after so many years of clinical studies there still seems to be no conclusive evidence to favour one over the other, is simply pointing prevention of Peri-implantitis being something that we cannot address in such a simplistic manner.
Peri-implantitis can be prevented by two things: proper design of the Implant Supracrestal Complex, combined with efficient oral hygiene. This is something we can achieve with both implant types, if we follow the proper design principles. And for each individual patient case, there might be advantages and disadvantages for each of the two implant types. The choice between bone or tissue level is important, but alone it will not make a difference. Thus, chose wisely based on the anatomy you are trying to recreate and the individual conditions, not on the basis of misplaced expectations.
Do you want to read more?
Click on the photo to read our new "Tissue Level vs Bone Level vol. 2" article, with all latest news and updates in this discussion!
Hear the latest news first: Subscribe to my newsletter!
References
Chan, D., Pelekos, G., Ho, D., Cortellini, P., & Tonetti, MS. (2019). The depth of the implant mucosal tunnel modifies the development and resolution of experimental peri-implant Mucositis: A case-control study. J Clin Periodontol, 46:248-255. doi:10.1111/jcpe.13066
Chappuis, V., Bornstein, M. M., Buser, D., & Belser, U. (2016). Influence of implant neck design on facial bone crest dimensions in the esthetic zone analyzed by cone beam CT: a comparative study with a 5-to-9-year follow-up. Clin Oral Implants Res, 27(9):1055-64. doi:10.1111/clr.12692
Derks J, Håkansson J, Wennström JL, Tomasi C, Larsson M, Berglundh T.J Effectiveness of implant therapy analyzed in a Swedish population: early and late implant loss.Dent Res. 2015 Mar;94(3 Suppl):44S-51S.
Katafuchi, M., Weinstein, B. F., Leroux, B. G., Chen, Y-W., & Daubert, D. M. (2018). Restoration contour is a risk indicator for Peri-implantitis: A cross-sectional radiographic analysis. J Clin Periodontol, 45(2):225-232. doi:10.1111/jcpe.12829
Mattheos, N., Vergoulis, I., Janda, M., & Miseli, A. (2021). The Implant Supracrestal Complex and its significance for long term successful clinical outcomes. Int J Prosthodont, 2021.
Fokas G, Ma L, Chronopoulos V, Janda M, Mattheos N. Differences in micromorphology of the implant-abutment junction for original and third-party abutments on a representative dental implant. J Prosthet Dent. 2019 Jan;121(1):143-150.
Mattheos N, Li X, Zampelis A, Ma L, Janda M.Investigating the micromorphological differences of the implant-abutment junction and their clinical implications: a pilot study. Clin Oral Implants Res. 2016 Nov;27(11):e134-e143.
Vianna, T. T., Taiete, T., Casarin, R. C. V., Giorgi, M. C. C., Aguiar, F. H. B., Silvério, K. G., Nociti Júnior, F. H., Sallum, E. A., & Casati, M. Z. (2018). Evaluation of peri-implant marginal tissues around tissue-level and bone-level implants in patients with a history of chronic periodontitis. J Clin Periodontol, 45(10):1255-1265. doi:10.1111/jcpe.12999
Monje, A., Caballé-Serrano, J., Nart, J., Peñarrocha, D., Wang, H-L., & Rakic, M. (2018). Diagnostic accuracy of clinical parameters to monitor peri-implant conditions: A matched case-control study. J Periodontol, 89(4):407–417. doi:10.1002/JPER.17.0454
Rokn, A., Aslroosta, H., Akbari, S., Najafi, H., Zayeri, F., & Hashemi, K. (2017). Prevalence of Peri-implantitis in patients not participating in well-designed supportive periodontal treatments: a cross-sectional study. Clin Oral Implants Res, 28(3):314-319. doi:10.1111/clr.12800
Yi, Y., Koo, K. T., Schwarz, F., Ben Amara, H., & Heo, S. J. (2020). Association of prosthetic features and Peri-implantitis: A cross-sectional study. J Clin Periodontol, 47(3):392-403. doi:10.1111/jcpe.13251
6 thoughts on “Bone level or Tissue Level..?”
Thank you Dr. Mattheos, Very nice sharing. Happy New Year!
Thank you for this very interesting and entertaining analysis!
One difference that I don’t think is arguable: The bone level implant is harder to take care of. You have to really get the floss down far under the gum line and wrap around that post. The tissue level? It’s just like any other tooth.
I have both – right next to each other – put in by the same oral surgeon. He never asked me if I’d prefer one vs the other. I wouldn’t have hesitated to pick the tissue level. Same brand of implant, I believe.
I just got back from the dentist. He had to remove a tiny bit of popcorn skin that was stuck under the bone level implant. Didn’t come out with floss or even after a cleaning! Down with bone level!
Thank you for your views! If I understand correctly, you are not a dentist (am I correct?), yet you are certainly very engaged with your implant treatment and you are very interested to know details, which I think is really commentable! Well done 🙂
Now with regards to the difference between bone and tissue level, I will not question your personal experience. Nevertheless, you have to also note that this is one case in a very specific anatomic condition (yours) which might not be something to generalise. Have you ever wondered why your dentist placed one bone level and one tissue level next to each other instead of using e.g. two tissue level? It was certainly not random choice. He probably saw something that was different between the two anatomic sites and made a choice. What you see as difference between two implants, might very well be the difference between two anatomic positions or prosthesis designs, one with very different limitations than the other. With regards to cleanability, no, I do not think that the choice of bone/tissue level makes any difference, it is rather the design of the prosthesis that matters. And wether a bone level or a tissue level is more suitable for the proper design, this depends very much on the individual anatomy of bone and soft tissues. In general, I believe the bone level allows for more options. With bone level I can do any design that is possible with tissue level. The opposite however is not true. Also, the depth of flossing is not depending on the type of implant, but again, the configuration of soft tissue and the prosthesis. This is a much deeper discussion that what a chatbox allows, so I hope it helps. I wish you a long life with both your implants healthy 🙂
Thank you for discussing this in such great detail, always enlightening to read your view. So how do you train your students? do you instruct them to use tissue level any more? or is bone level enough to know for all indications?
thank you for that, an interesting question indeed! We start the training by teaching the fundamental principles of technology and biology. Then when it comes to applying these principles with a product, my usual strategy is to use one implant only for first 15-20 cases. After that the student is starting to master the workflow and feel confident with procedures and compoenents and then is the time to increase complexity and introduce more complex components. The aim is to have an independent and comppetent clinician at the end of the training who can handle complex cases and select the most appropriate system and components. The Tissue Level implants are in my mind ideal for beginners, well suited for straightforward cases, simpler “cookbook” workflow and fewer components. Once this level is mastered one can advance to more complex bone level implants with different designs and more versatile osteotomy protocols and options for more individual componentry.